Peter Kolchinsky, PhD
This is the first in a series of articles that aim to define the biopharmaceutical industry’s social contract with America, to examine practices that deviate from that contract, and to propose refinements to healthcare policy to ensure that our continued investment in scientific progress ultimately yields affordable, effective therapeutics for future generations.
Article 1: America’s Social Contract with the Biopharmaceutical Industry
Article 2: What happens when a drug won’t go generic?
Article 3: Protecting Off-Patent Sole-source Drugs from Price-Jacking
Please see Important Disclosures for Readers at the end.
The public has long voiced outrage at pharmaceutical companies over high drug prices. Many patients feel the real pain of rising healthcare costs and frequently call for price controls on prescription drugs. But price controls are not the solution. In fact, high prescription drug prices aren’t even the problem.
Miscast as a bloated expenditure, high prices on branded drugs incentivize and attract society’s talent and capital to the biopharmaceutical industry to fund and research new cures and treatments that will eventually become inexpensive generic drugs. These resulting generics are one of humanity’s most valuable and underappreciated resources.
High branded-drug prices are necessary to grow this mountain of inexpensive generic drugs. At the same time, we can’t allow anyone’s child or aging parents to be denied the medication they need on account of cost. The reason why new drugs seem unaffordable lies not with branded drug prices but with the intentionally poor design and distribution of healthcare insurance in our country. Branded drugs can be made more affordable — without imperiling biopharmaceutical innovation — by ensuring that insurance properly covers patients, sparing them the euphemism of cost sharing (e.g. co-pays and deductibles).
Besides shielding patients from drug costs and improving immediate health outcomes, this proposal will drive the scientific innovation needed to build a legacy of inexpensive treatments for future generations. Alongside other grand objectives like universal access to clean water and energy efficiency, building this mountain of useful generic drugs as rapidly as possible should be a national and global priority, one that America has led and should continue to lead. If we stall this engine of progress through price controls or other blunt measures meant to ease costs today, we condemn the future to be no better than the present. In turn, the biopharmaceutical industry should do its part to ensure all branded drugs are eventually and adequately genericized.
An underappreciated resource
 While high-priced branded drugs make up only 11% of all prescriptions¹, they are the source of most pharmaceutical revenue ($237 billion in 2015²). Typically after 10–15 years, branded drugs lose their patent protection, unleashing low-cost generic versions³ (in 2015 the US spent a comparatively modest $88 billion on generics, though these drugs make up 89% of all prescriptions⁴). Unlike almost any other aspect of healthcare, drugs constantly go generic, which keeps total spending on retail drugs around a mere 12% of total healthcare costs, about the same as it was in 1972.⁵ And even though drug spending remains the same percent of overall healthcare spending, we now have far more and better treatments.⁶
While high-priced branded drugs make up only 11% of all prescriptions¹, they are the source of most pharmaceutical revenue ($237 billion in 2015²). Typically after 10–15 years, branded drugs lose their patent protection, unleashing low-cost generic versions³ (in 2015 the US spent a comparatively modest $88 billion on generics, though these drugs make up 89% of all prescriptions⁴). Unlike almost any other aspect of healthcare, drugs constantly go generic, which keeps total spending on retail drugs around a mere 12% of total healthcare costs, about the same as it was in 1972.⁵ And even though drug spending remains the same percent of overall healthcare spending, we now have far more and better treatments.⁶
Each year, the biopharmaceutical industry produces new drugs that raise our collective health, give hope to patients suffering from debilitating diseases, and further our understanding of human biology. Because the underlying biological processes inside our cells and bodies are essentially unchanging, the drugs we use today will work just as well in a hundred years, with few exceptions (e.g. antibiotics become obsolete and require replacement as bacteria evolve to become resistant). Many drugs may even work better with improved diagnostics, delivery technologies, and insight into how to best combine therapies. Therefore, much of the scientific progress we achieve in our lifetime will also accrue to the benefit of future generations.
Generics, which the FDA ensures work just as well and meet the same safety standards as original branded drugs, offer huge cost savings within months of coming to market and, with few exceptions, remain inexpensive for the rest of time.⁷
We need only look to the past to understand what progress can be made. Too often, we discount the ingenious tools for preventing, treating, and curing diseases that have made America and the whole world richer.⁸ Over the last hundred years, advancements in medicine, particularly perinatal care, vaccines, and antibiotics, as well as hygiene and nutrition contributed to the nearly 30 years added to the average American’s life expectancy.⁹ Over the same period, global life expectancy doubled.¹⁰ Quality of life also improved with groundbreaking treatments for heart disease, pain, diabetes, and many
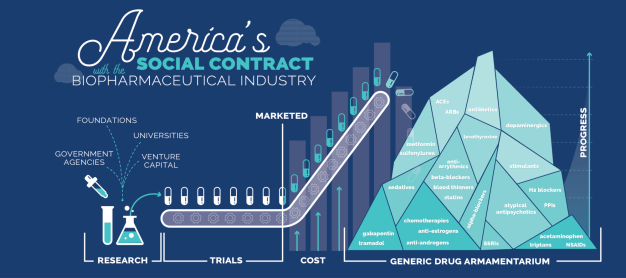
 Cholesterol-lowering statins alone have contributed to the 50% decrease in death from both heart attacks and strokes throughout the developed world over the last few decades. When Pfizer’s patent on the best-selling statin Lipitor expired and generic versions entered the market, the price quickly dropped by 95% of the initial cost and has remained comparatively negligible ever since.¹² Gleevec, a highly successful drug that restores many years of life to patients suffering from chronic myelogenous leukemia (CML) and other cancers, has also recently gone generic, resulting in its price trending steadily downward. Today, our generic drug armamentarium includes treatments that manage blood pressure (ACEs, ARBs), cholesterol (statins, fenofibrate), diabetes (metformin, insulin), migraines (triptans, acetaminophen, NSAIDs), inflammation (steroids, methotrexate, 5-ASAs), chronic pain (gabapentin, tramadol), depression (SSRIs), schizophrenia (atypical antipsychotics), heartburn (H2 blockers, PPIs), hypothyroidism (levothyroxine), infections (antibiotics), enlarged prostate (alpha-blockers), insomnia (sedatives), ADHD (stimulants), Parkinson’s (dopaminergics), and cancers (anti-estrogens, anti-androgens, chemotherapies), as well as treatments that prevent heart attacks and strokes (blood thinners, anti-arrhythmics, beta-blockers). These represent just a glimpse of the medicines that biopharma can offer to humanity in this century if we continue to fuel and incentivize this industry.
Cholesterol-lowering statins alone have contributed to the 50% decrease in death from both heart attacks and strokes throughout the developed world over the last few decades. When Pfizer’s patent on the best-selling statin Lipitor expired and generic versions entered the market, the price quickly dropped by 95% of the initial cost and has remained comparatively negligible ever since.¹² Gleevec, a highly successful drug that restores many years of life to patients suffering from chronic myelogenous leukemia (CML) and other cancers, has also recently gone generic, resulting in its price trending steadily downward. Today, our generic drug armamentarium includes treatments that manage blood pressure (ACEs, ARBs), cholesterol (statins, fenofibrate), diabetes (metformin, insulin), migraines (triptans, acetaminophen, NSAIDs), inflammation (steroids, methotrexate, 5-ASAs), chronic pain (gabapentin, tramadol), depression (SSRIs), schizophrenia (atypical antipsychotics), heartburn (H2 blockers, PPIs), hypothyroidism (levothyroxine), infections (antibiotics), enlarged prostate (alpha-blockers), insomnia (sedatives), ADHD (stimulants), Parkinson’s (dopaminergics), and cancers (anti-estrogens, anti-androgens, chemotherapies), as well as treatments that prevent heart attacks and strokes (blood thinners, anti-arrhythmics, beta-blockers). These represent just a glimpse of the medicines that biopharma can offer to humanity in this century if we continue to fuel and incentivize this industry.
The collective armamentarium of branded drugs will mostly be generic within 10–15 years and will add to our high-value, low-cost arsenal. These include better treatments for many of the conditions mentioned above as well as breakthrough therapies or cures for cystic fibrosis, HIV, Hepatitis C, and many cancers. Yet so many problems remain to be solved. We must discover how to treat and cure diseases that run from uncomfortable to excruciating to devastating. Initially expensive, these drugs will soon enough go generic and join our growing mountain of cost-effective medications.¹³
Branded drugs are worth the cost
Branded drug expenditures represent 1.8% of the US GDP.¹⁴ That’s a little more than a tenth of total healthcare spending and less than the 2.4% of GDP spent by the US government on roads, drinking water, and wastewater infrastructure, which few dispute as necessities worthy of investment.¹⁵ But because health insurance plans, including Medicare, engage in aggressive cost-sharing requiring co-payments as high as 20% of the price of a drug, vulnerable patients disproportionately carry the burden of building our common stockpile of generic treatments.¹⁶
Increased infrastructure investment stands as one of the last few areas of clear bipartisan agreement.¹⁷ We know that we need functional roads and water and sewage treatment facilities. We may even know that infrastructure spending will result in more jobs when Americans fix our roads and bridges.¹⁸ And yet, all this holds true for the drug industry: we all will eventually need what the millions of people employed directly or indirectly by the biopharmaceutical sector can create for us.
High-priced branded drugs power this entire biopharmaceutical ecosystem.¹⁹ Drug price controls would imperil it, signaling that some or even all the people involved should do something else for society, perhaps work in a different industry. Therefore, before we accidentally hobble or dismantle the biopharmaceutical industry in an effort to make today’s high-priced drugs cheaper as quickly as possible, we need ask whether branded drugs are “worth it” — essentially whether their price matches their value.
To understand a drug’s true value, consider each person that it will help as a branded drug in the present and as a generic in the future. Medicine has influenced every dimension of our lives, contributed to the advancement of art and technology that permeates our civilization by allowing innovators to live longer and healthier lives, and impacts our social fabric in immeasurable ways. Even a modest medical breakthrough that will someday go generic offers society high value for low cost.²⁰ We can collectively afford to make this investment.
This promise to create a mountain of low-cost therapies forms the core of the compact between drug developers and patients, which is to say all of us. Social contracts can be explicit, such as when Americans gave their representatives the power to govern them within the democratic precepts of the Constitution and the Declaration of Independence. In other instances, social contracts become implicit agreements between company and consumer. Google’s memorable motto “Don’t be evil” (now “Do the Right Thing”) became an implied promise to the user that “in exchange for using our services, we won’t do anything monstrous with the power you’ve given us.” Biotech operates on its own social contract with the American patient: “Yes, prices will be initially high on branded drugs. But after a patent expires, you enjoy the benefits of the cheap, effective generic versions for the rest of time.”
Others have proposed the notion of a social contract, notably with emphasis on keeping branded drug price increases in check while they are patent-protected.²¹ But as long as a drug ultimately will go generic, it’s not all that critical to the long-run price/value assessment whether it starts off at a lower price, climbs, and then goes generic, or if it starts off at a higher price, stays flat, and then goes generic.
Genericization without undue delays is the only critical feature of the contract. Consider the biopharmaceutical industry as putting novel branded drugs on a conveyer belt that over 10–15 years transports them to the top of the generic drug mountain– that final moment when the drug goes generic and falls off the conveyer onto the eternal generic mountain is the ultimate fulfillment of the industry’s end of the social contract.
There is more work to do in this regard. In the case of Gleevec, its price has been floating gently down after it went generic rather than fallen off a cliff, as Lipitor’s did. FDA approval of more generics would help speed the fall. In the case of biosimilars — which are as close as we can scientifically come to making a generic version of complex drugs such as antibodies and enzymes that are manufactured in biological systems — we are only just beginning to witness and assess the market dynamics around adoption and price erosion. But considering the cost of biologics production is in most cases roughly 5–15% of a biologic drug’s price, biosimilars may eventually be sold for at least a 70% discount. Companies will pursue every legal means at their disposal to prevent the uptake of biosimilars, as we have seen many innovators do to forestall generic entrants.²² The generics industry and the Federal Trade Commission estimate that America is losing out on almost $10 billion of savings each year due to such stall tactics, which is a problem and makes for many galling headlines, but it’s also less than 3% of total drug spend²³. Arguably the industry as a whole can afford to solve this public relations headache by swearing off excessive cleverness, and regulators should pursue avenues to accelerate genericization without impacting innovation, but regardless, those stall tactics fail in the long run and prices fall as competition sets in, as they have been steadily falling for Gleevec and its generics. For the few drugs where no true generic could ever exist — say, in the case of certain cell or gene therapies — new pricing mechanisms for the synthetic genericization of such drugs can and should be created, adapting pricing schemes already in place for pandemic vaccines and other biodefense products.
If we agree that the high prices of branded drugs legitimately drive investment towards society’s ownership of more and better generics, then we also need to condemn those who steal generics away from society. Making sure that drugs go generic and thereafter remain inexpensive is the long-term value side of the social contract. Therefore, when a company takes a formerly inexpensive generic and raises the price well above the cost of production without improving the drug, that company violates the contract. Several companies have recently exploited loopholes that regulators should seek to close quickly, for example by accelerating the approval of more generics to keep prices down and making sure that generic manufacturers can access samples of an original drug to help them demonstrate the equivalence of their generic product, both of which the FDA has vowed to do.²⁴
Imagine though, that all new drug development stopped, and all current branded drugs immediately went generic, for example through price controls or by invalidating their patents. Total drug spending would fall by approximately 70%, resulting in a one-time 9% reduction in healthcare spending, equating to about 1.5% of US GDP. Within a few years though, healthcare costs would revert to previous levels due to the rising costs of hospitals, surgeries, and other non-genericizable services.²⁵ Meanwhile, our mountain of generic drugs would cease growing — the conveyer belt will grind to a halt. Therefore, in exchange for a modest and temporary savings in our collective budget, we would destroy biopharma’s innovative core that, as of this moment, is advancing new treatments for migraines, bone fractures, cancer, diabetes, and countless other diseases that send patients, young and old, to hospitals each year. As overall costs continued to grow, the public would realize what Congress and insurance companies already know: the bulk of healthcare costs stem from hospitals and services, none of which will ever go generic.²⁶
A call to end cost-sharing
Some argue that the biopharmaceutical social contract places responsibility on drug companies to make sure that patients have access to their drugs²⁷. This stance ignores the role of payers, private insurance companies as well as Medicare, in implementing high co-pays and other cost-sharing mechanisms to discourage patients from using medications that their physicians prescribe. The cost of most branded drugs could be cut in half and the majority of people in America still couldn’t afford to pay Medicare’s required 20% co-pay.
The tactics companies employ to try to make sure that patients can afford their share of the cost of a drug, such as co-pay assistance, are sometimes portrayed as bribes that companies pay patients to take their medications. Payers may worry that a pharmaceutical company has inappropriately marketed a drug, encouraging physicians to prescribe it to patients who don’t need it. When this happens, the government should prosecute and punish companies, but it’s inappropriate to cast suspicion on every doctor-patient interaction that results in a prescription for a high-priced drug and then, via cost-sharing, coerce patients into second-guessing their physicians. Many companies have programs to provide free drug to patients without insurance. And yet, many patients don’t realize that such programs exist, assume that they won’t be able to afford a drug, and don’t bother getting a prescription filled. Physicians are often too busy to explain the details of co-pay assistance to each patient. These barriers to access create their intended result: fewer patients fill prescriptions that their physicians write.
We have also entrusted our government and insurance companies to distribute the costs of healthcare across our population and across time so that people unfortunate enough to be sick don’t suddenly find themselves choosing between the branded drugs they need and their mortgage payments. Insurance practices in America conform to laws passed by our elected representatives. So if the politicians we elect to Congress allow insurance companies to turn away people based on pre-existing conditions or to charge high co-pays, then we have harmed ourselves.
A common complaint is that healthcare is more expensive in the US than anywhere else in the world but that our outcomes lag far behind.²⁸ This has little if anything to do with pharmaceuticals. On the contrary, the US enjoys faster approvals of innovative drugs, giving many US patients access to important medicines, such as cures for hepatitis C, years before they are used widely in Europe.²⁹ At the same time, the US makes the most use of generic drugs — more than any major healthcare market. While 89% of prescriptions filled in the US are for generics, throughout Europe generic utilization remains far lower, from Switzerland (17%) to France (30%) to Spain (47%) to Germany (80%).³⁰ Therefore, although Europeans pay less than Americans for branded drugs initially, they pay branded prices for longer because they don’t switch to generics as quickly as possible. As much as it is popular to cite how much more the US pays for a new drug, there are also many examples of older drugs for which Europe pays more. While the US has already cured many patients suffering from hepatitis C, for example, Europeans are spending more money on older, less effective treatments.³¹ Despite having the most advanced technologies, the US system, through cost-sharing, non-comprehensive insurance, and bureaucracy, makes healthcare unaffordable and practically inaccessible for some, which then weighs on healthcare outcomes for these individuals and also shows up in national statistics.³²
Copays and deductibles imposed upon patients force many to stop taking their medications as prescribed, leading to worse outcomes and higher long-term costs. Even small co-pays and fees — as low as $1 to $5 — reduce drug adherence and cause patients to avoid necessary care.³³ Approximately 50% of patients fail to adhere to their prescriptions due in part to high co-pays, while high deductibles push patients to cut out valuable preventative services. Greater cost-sharing is also associated with increased emergency room visits.³⁴ Low-income patients are often the most vulnerable. Indeed, the very term “cost-sharing” disguises what could be more aptly described as an institutionalized failure to insure.
If we truly hope to expand access and unburden patients of the financial hardship of getting sick, then rather than blaming the innovators for the prices of their newest drugs, let’s upgrade our insurance system to one that actually provides proper insurance to everyone. Spending should be spread more evenly instead of hitting patients with toxic cost-sharing when they are sick and most vulnerable. Cost-sharing just adds insult to injury. Imagine if, after being mugged, the victim had to make a co-pay to have the police show up.
Cutting out-of-pocket costs would lead to improved adherence, better health outcomes, and lower long-term healthcare spending, provided that physicians do not encourage patients to over-utilize an “all you can eat” healthcare system. While this is an important caveat, it does not justify cost-sharing, which shifts the responsibility of setting an appropriate level of care from knowledgeable physicians to distressed patients. Co-pays and deductibles are intended to make sure that individuals have some “skin in the game” and think twice before seeking unnecessary treatments. However, few patients can tell the difference between a necessary and unnecessary treatment when the person prescribing the treatment is a doctor.
If a doctor prescribes an expensive branded drug despite the availability of generics or less expensive but similar therapies, it makes sense to nudge patients towards switching. The problem is when payers try to discourage patients from taking uniquely beneficial drugs for which there are no substitutes. Behavioral economists conceded long ago that humans do not always behave rationally, often undervaluing their future selves. When insurers place significant cost burdens on a patient, they nudge individuals to indiscriminately reduce their use of both low-value and high-value treatments and services, often with costlier downstream consequences.
Specific private and government insurance practices were designed with high co-pays, maybe thoughtlessly or maybe heartlessly, to make patients believe that drugs are unaffordable. Yet, the 1.8% of GDP that America spends on branded drugs does not have to feel unaffordable to any of the people who need these medications, just as our nation’s road and drinking water infrastructures are considered a basic resource available to all.
Insurance companies should address overutilization of services and unnecessary prescriptions by confirming that physicians adhere to standard of care guidelines, not by underinsuring patients so they are financially discouraged from following their doctors’ advice. And insurers should align physician incentives with the quality of care rather than the quantity of care. Fortunately, there are many ongoing experiments being run by hospitals, physician networks, and insurers to figure out how to do just this.³⁵
A ‘fair price’ conundrum
Insurance only serves its function if everyone, especially healthy people, pays what they can. Just as taxes grow in proportion to income, insurance companies should ensure that an individual pays their highest rates during their highest income years, which also happens to be when they are younger and most likely to be healthy. It makes little sense to increase costs on people when they are older, have less income, and yet are most likely to need healthcare. The young, healthy, working-age population might not like paying more, but they will eventually find themselves in need of the medicines whose development they helped fund while they were younger. Until insurance functions properly, patients driven to desperation by cost-sharing will understandably find government-imposed drug-price controls appealing, regardless of the consequences to the biopharmaceutical ecosystem.
There has always been difficulty establishing a drug’s cost-effectiveness, a quantification of whether it is “worth it”. One can make the case that a patient with elevated cholesterol who skips treatment to save money will only end up costing himself, his family, and society more if he later suffers a heart attack, and that, therefore, the patient and society should rationally want the patient to get preventative treatment. However, the cold hard math of dollars spent and saved does not always support this conclusion because it depends on the cost of the treatment and the value one places on life. An oft-cited macabre bit of trivia is that smokers save taxpayers’ money on Medicare and Social Security by dying more quickly and earlier than non-smokers. If it were all about money, we should be encouraging smokers to keep lighting up. But we are using a calculus that heavily weighs well-being, which basically means that the benefits of healthcare are unquantifiable, at least in America. As a society, we have not come to terms with how to put a price on life (one could argue that the EPA sets the value of a statistical life at $10M in 2016, FDA just under at $9.5M, and the Department of Agriculture at $8.9M³⁶) and seem to believe that paying whatever it takes to treat or prevent pain, disability, and death is always the “right” choice, or at least the American choice. Anyone who puts money over life is considered heartless.
Yet we know that our inability to say “no” to effective medications on the basis of cost makes us vulnerable to price gouging, and we know that drug companies know this, calling into question whether drug companies are charging a “fair” price, even as we struggle to define what fair is. Some would argue that fair means pegging prices to the cost of drug development, calling for every company to disclose what it cost them to develop each drug. Others suggest it’s only fair to charge for a drug if it works, although in practice we often do not know if a drug is working for a specific patient.
Neither of these suggestions would be impossible to implement in some form and, through transparency, might even help make drug development more efficient. The drug industry’s profit margins are roughly 20%, which some say are too high. But we know that there isn’t room to reduce branded drug prices by half, for example, at least not without cutting salaries and laying off people. There are no doubt inefficiencies in every company, but which specific expenses are unnecessary is debatable. Profit margins appear modest for an industry accused of being a price gouging monopoly.
There is a good chance that, after every R&D expense has been audited by Congress and when companies are only allowed to charge for drugs that work, the industry will be found to be justified in charging a collective 1.8% of US GDP for branded prescription drugs. Where would the public outrage then turn? Maybe it will finally turn where it needs to focus, on the lack of universal insurance in America and the heartless practice of imposing cost-sharing on the sick and vulnerable. Inadequate insurance coverage and high co-pays are at the root of why patients go without. Insurance companies and government policies are standing in the way of patients accessing medications that could help them.
There would be less outrage if patients could count on their insurance plans to pay in full for whatever drug their physicians prescribed. Controlling the prices of the more expensive branded drugs is counter-productive and, in the long run, irrelevant, as long as those drugs will go generic.
The central element of the biopharmaceutical social contract, as we see it, has little to do with what price is fair for a branded drug. That’s an unwinnable debate and a search for unquantifiable value. Instead ask whether or not a drug will eventually go generic, offering society reassurance that there is an end in sight to the high cost of any new drug. Basically, whatever the branded price, that high cost is finite, whereas the value society will enjoy is infinite. Some might think that hepatitis C drugs approved since 2015 were indisputably overpriced, yet when the first hepatitis C drugs came to market, the public outcry over their high cost for a short course of treatment was hardly universal. Many articles pointed to how much more cost-effective these curative drugs were compared to previous HCV therapies or HIV medications that cost less but must be taken chronically. And the more productive the biopharmaceutical industry has become, the more likely it is that multiple competitors come to market with comparable drugs within a few years of one another, spurring some price competition even before generics set in, as has happened with treatments for hepatitis C, diabetes, and cholesterol management. It’s increasingly rare for any drug company to enjoy a monopoly for very long.
Indisputable price-gouging, like price-jacking an old generic drug, does happen, but on a comparatively small scale. These practices can and should be eliminated (the FDA has already taken steps to do so by approving generics more quickly). There may even be a role for price controls by having the government contract with manufacturers of old single-source generics (i.e. those supplied by just one company, usually because the residual demand for these older drugs is too small to sustain multiple competitors), ensuring a continuous supply at a modest profit margin.
Populist demands to cut drug prices may tempt some, but the innovative edge of the pharmaceutical industry is powered by the willingness of the US market to reward groundbreaking research. Even European pharmaceutical companies would likely not bother investing in the development of many kinds of new drugs if they couldn’t count on selling them in the US. Hitting the biopharma industry with price controls in reaction to understandable but misdirected outrage would only rob ourselves and our children of the compounded growth of our giant mountain of generic drugs. If we conflate investing in new drugs that we will someday “pay off” with simply spending money on expensive healthcare services (which never go generic), then we may make the wrong budget cuts and thereby only worsen our long-term costs and outcomes.
Consider a home mortgage. A borrower spends 15–30 years paying off a mortgage, typically while younger and employed. Those mortgage payments can be substantial, but once the house is paid off, it is lived in rent free and then passed down to children and grandchildren. By comparison to drugs, doctors, surgeries, and treatments such as dialysis are pure rent and will remain expensive forever — we can only hope to prevent the need for these expensive services with inexpensive drugs.³⁷ A heart bypass and a cardiac surgeon may never go generic, but cholesterol lowering statins, which prevent hospital admissions and even surgeries, will be cheap indefinitely.
When President Kennedy proposed that Americans send a man to the moon, he asked the country to boldly commit to a goal that would require substantial funding, partnerships between government and corporations, and immense intellectual capital. He explained that “in a very real sense, it will not be one man going to the moon — if we make this judgment affirmatively, it will be an entire nation. For all of us must work to put him there.”³⁸ Individual scientists and research teams will be the ones to discover the next generation of cures and treatments, but their successes will be the product of a collective societal effort. Just as the Apollo Program produced breakthroughs in engineering, computing, and our understanding of spaceflight, let us recognize that the high cost of today’s new drugs fuels and inspires the continued discovery of tomorrow’s medical advances, providing high-value, affordable generics that will benefit us all for the rest of time.
Acknowledgements: I’m grateful to Aaron Hiltner and Chris Morrison for their invaluable and substantive thought-partnership and drafting/editing and to everyone who engaged with me in the constructive debates that led up to this article.
Article link: https://medium.com/the-biotech-social-contract/kolchinsky-tbsc-1-dafc2fe803e5
Sources
¹ http://www.gphaonline.org/media/generic-drug-savings-2016/index.html.
² https://www.cdc.gov/nchs/data/hus/hus16.pdf#094; http://www.gphaonline.org/media/generic-drug-savings-2016/index.html.
³ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4899342/.
⁴ http://www.gphaonline.org/media/generic-drug-savings-2016/index.html.
⁵ https://data.oecd.org/healthres/pharmaceutical-spending.htm; https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/downloads/highlights.pdf.
⁶ http://content.healthaffairs.org/content/23/1/10/F1.expansion.html; https://www.cdc.gov/nchs/data/hus/hus16.pdf.
⁷ http://www.nejm.org/doi/full/10.1056/NEJMp1704907?af=R&rss=currentIssue#t=article.
⁸ https://data.oecd.org/healthstat/potential-years-of-life-lost.htm;
https://data.oecd.org/healthstat/life-expectancy-at-birth.htm#indicator-chart.
⁹ http://u.demog.berkeley.edu/~andrew/1918/figure2.html; http://gamapserver.who.int/gho/interactive_charts/mbd/life_expectancy/atlas.html; http://jamanetwork.com/journals/jamainternalmedicine/article-abstract/2626194; https://ourworldindata.org/life-expectancy/.
¹⁰ http://gamapserver.who.int/gho/interactive_charts/mbd/life_expectancy/atlas.html; https://ourworldindata.org/life-expectancy/.
¹¹ http://u.demog.berkeley.edu/~andrew/1918/figure2.html; http://gamapserver.who.int/gho/interactive_charts/mbd/life_expectancy/atlas.html; http://jamanetwork.com/journals/jamainternalmedicine/article-abstract/2626194; https://ourworldindata.org/life-expectancy/
¹² https://hbr.org/2014/11/the-real-cost-of-high-priced-drugs
¹³ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4899342/
¹⁴ The most recent figures come from 2015. In 2015, the U.S. spent $325B on retail drugs and an estimated $128B on non-retail drugs, for a total of $453B. For retail drugs, branded drugs made up 73% of all spending. If we apply the same estimate to non-retail drugs, we see that the U.S. spent $331B on branded drugs. The U.S. GDP was $18.0366T in 2015, leading to a calculation of 1.8%. https://www.cdc.gov/nchs/data/hus/hus16.pdf#094; https://aspe.hhs.gov/system/files/pdf/187586/Drugspending.pdf; http://www.gphaonline.org/media/generic-drug-savings-2016/index.html.
¹⁵ From the above footnote we see that the U.S. spent $331B on branded drugs. The U.S. spent $3.2056T on healthcare. $331B/$3.2056T = 10.3%. See https://www.cdc.gov/nchs/data/hus/hus16.pdf#094. For GDP spent on roads, drinking water, and wastewater infrastructure see https://www.cbo.gov/publication/52463.
¹⁷ http://i2.cdn.turner.com/cnn/2017/images/03/08/rel4d.-.budget.pdf.
¹⁹ http://www.gphaonline.org/media/generic-drug-savings-2016/index.html.
²⁰ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4899342/.
²¹ https://www.allergan.com/news/ceo-blog/september-2016/our-social-contract-with-patients.
²² https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4899342/
²³ http://www.gphaonline.org/media/cms/REMS_Studyfinal_July2014.pdf; https://www.ftc.gov/sites/default/files/documents/reports/pay-delay-how-drug-company-pay-offs-cost-consumers-billions-federal-trade-commission-staff-study/100112payfordelayrpt.pdf
²⁵ https://data.oecd.org/healthres/health-spending.htm; https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/Downloads/Proj2015.pdf.
²⁸ http://www.nejm.org/doi/full/10.1056/NEJMp1708704
²⁹ http://www.who.int/intellectualproperty/events/en/R&Dpaper2.pdf; http://eprints.lse.ac.uk/71641/7/Wouters_Comparing%20generic%20drug%20markets_2017_author%20LSERO.pdf
³⁰ http://www.gphaonline.org/media/generic-drug-savings-2016/index.html; http://eprints.lse.ac.uk/71641/7/Wouters_Comparing%20generic%20drug%20markets_2017_author%20LSERO.pdf
³² http://www.commonwealthfund.org/publications/in-the-literature/2016/nov/2016-international-health-policy-survey-of-adults; http://www.nejm.org/doi/full/10.1056/NEJMp1708704
³⁴ https://jamanetwork.com/journals/jama/fullarticle/1108322
³⁵ http://catalyst.nejm.org/simple-case-health-insurance-complexity/ ; http://www.choosingwisely.org/
³⁶ https://www.bloomberg.com/graphics/2017-value-of-life/
³⁷ http://www.nbcnews.com/health/health-care/surgery-prices-surge-innovation-consolidation-under-obamacare-n191901 ; https://hbr.org/2014/11/the-real-cost-of-high-priced-drugs
³⁸ https://www.nasa.gov/vision/space/features/jfk_speech_text.html
Peter Kolchinsky, PhD
Peter Kolchinsky is a founder, Portfolio Manager, and Managing Director at RA Capital Management. Peter is active in both public and private investments in companies developing drugs, medical devices, diagnostics, and research tools, and serves as a Board Member for various public and privately held companies, including Dicerna Pharmaceuticals, Inc. and Wave Life Sciences Ltd. Peter also leads the firm’s outreach and publishing efforts, which aim to make a positive social impact and spark collaboration among healthcare stakeholders, including patients, physicians, researchers, policy makers, and industry. He authored “The Entrepreneur’s Guide to a Biotech Startup” and served on the Board of Global Science and Technology for the National Academy of Sciences. Peter holds a BS from Cornell University and a PhD in Virology from Harvard University.
Important Disclosures for Readers
The opinions in this article are my own, subject to change, and may not reflect the opinions of RA Capital Management or anyone with whom I am affiliated. With regard to data and anything presented as fact, I’ve made an effort to be accurate and cite sources I believe to be accurate, but it’s possible I’ve erred or that my sources were inaccurate, in which case hopefully not in ways that materially impact the overall argument. If you think you’ve spotted an error, please contact me to help me improve my reasoning. While you probably already know this, regulations I’m beholden to as an investor nonetheless require me to remind you that nothing may turn out the way I want things to or predict that they might in this article, whether with a company, the healthcare system, or the economy in general. This article may reference specific companies to provide real-world examples of otherwise generalizable abstract concepts; nothing in this article is intended to be taken as investment advice in any company or in my firm. To help you judge my conflicts of interest, let me draw your attention to the possibility that I or my firm may have invested, may be invested, or someday may invest in the securities of companies mentioned in these articles. If I or my firm have a relationship or investment position in any company mentioned in an article, that would be specifically disclosed in the text of the article at the time of publication and will be accurate as of the time of initial publication but may change thereafter.


 When either updating an old RVU or coming up with a new one, the RUC members spend most of their time debating something called “work units”—a slippery currency that combines how much time, training, technical skill, physical and mental effort, and stress are required of a doctor when performing a certain procedure or service. For example, according to the 2013 RUC database, “freezing off” a suspicious-looking lesion or freckle, known in the medical parlance as a “destruction,” has been assigned 1.22 work units; inserting a single-valve cardiac stent, 33.75.
When either updating an old RVU or coming up with a new one, the RUC members spend most of their time debating something called “work units”—a slippery currency that combines how much time, training, technical skill, physical and mental effort, and stress are required of a doctor when performing a certain procedure or service. For example, according to the 2013 RUC database, “freezing off” a suspicious-looking lesion or freckle, known in the medical parlance as a “destruction,” has been assigned 1.22 work units; inserting a single-valve cardiac stent, 33.75. Another flaw in methodology comes from the fact that the RUC often relies on records from teaching hospitals in determining how long an operation takes, even though teaching hospitals often have longer surgery times than nonteaching hospitals. A 2006 study by the nonprofit health care research firm RTI International compared the amount of time the RUC suggested for sixty surgeries to data from 148 hospitals’ actual surgery logs. The RUC’s estimated times were often longer—sometimes by up to two hours.
Another flaw in methodology comes from the fact that the RUC often relies on records from teaching hospitals in determining how long an operation takes, even though teaching hospitals often have longer surgery times than nonteaching hospitals. A 2006 study by the nonprofit health care research firm RTI International compared the amount of time the RUC suggested for sixty surgeries to data from 148 hospitals’ actual surgery logs. The RUC’s estimated times were often longer—sometimes by up to two hours.


 Over 16 million text messages are sent in one internet minute in 2017.
Over 16 million text messages are sent in one internet minute in 2017.